
- +1 (714) 578-6100
Hours Mon - Fri, 07:00 AM - 06:00 PM (Pacific Time)
Vacuum Overview
“Vacuum” means the complete absence of matter. In order to create vacuum conditions, pressure within a given space must be lower than the surrounding environment. The quality of the vacuum is dependent upon several factors, including the number of particles within a given space. The inertia of moving particles exerts pressure, so fewer particles means lower pressure. Scientists have given up on the idea that a perfect vacuum is even possible; space is the most ideal vacuum we know of, but hydrogen atoms, energy waves and other moving particles are still present.
Vacuum Chambers
Imperfect as they are, we create vacuums for various applications and processes: sealed chambers that help researchers and manufacturers with tasks including leak testing, stress testing, degassing, drying, distillation, permeability testing, coating, specific gravity determination,
Right on the heels of a blog published on Laboratory-Equipment.com called “How to Improve Pipetting Techniques,” we present this discussion about calibration of gravimetric pipettes. Most labs are required to complete periodic calibration of instruments, as per documented protocols. Whether the tasks are performed by lab staff or a vendor, it’s a time-consuming and exacting process. The alternative? Data and test results that may be questioned or challenged.
Considerations for Calibration
Pipette calibration is performed by laboratories to ensure that their pipettes are functioning within given (documented or established) parameters. In fact, pipettes that are not routinely calibrated can produce vastly different dispensing volumes than expected, which results in ambiguous or even erroneous data sets.
Seems like a cleanroom is no place for a ladder: a dirty, dusty, paint-smeared structure used by the contractor who does the termite repair on your house. But in fact, cleanrooms are perfectly-suited to ladders, if they are Terra’s BioSafe brand. Cleanroom and lab ladders are used to access out-of-reach filters, lighting, or equipment. For example, taking particle measurements and airflow readings below the filter face in preparation for an ISO inspection may require a ladder. And what about changing light bulbs and making small repairs? Terra’s shelf platform can be attached to ladders, providing a convenient surface for tools and small instruments.
 BioSafe folding cleanroom ladders have a number of unique features that make them ideal for aseptic
BioSafe folding cleanroom ladders have a number of unique features that make them ideal for aseptic
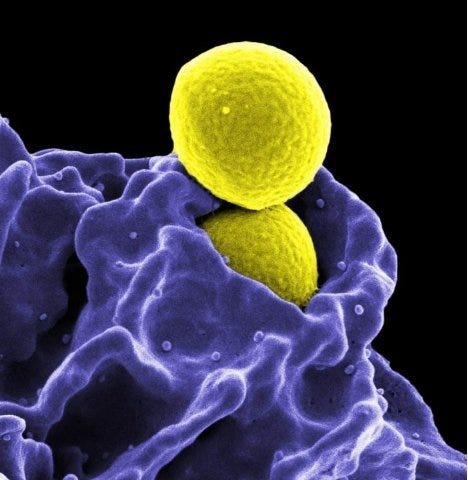
Methicillin-Resistant Staphylococcus aureus (MRSA) bacterium. Photo credit: CDC
Controlling microbial contamination is one of the leading concerns in research, clinical, and medical facilities. Microorganisms (hazardous or not) can put personnel, patients and caregivers at risk. In hospital and medical facilities, patients are often immuno-compromised or have serious conditions that make them particularly susceptible to opportunistic microbes or secondary infections.
For these reasons, many products are available for decontamination of these critical spaces. There are differences in product effectiveness, cost, potential residual damage and operational limitations. Two particular methods, presented here, are commonly employed to reduce or eradicate hazardous microorganisms.
Ultraviolet germicidal irradiation has been a mainstay for killing and inactivating microorganisms
Flammable gasses and vapors possess the potential to cause devastating damage to personnel, property and the environment. To minimize this danger, organizations including Occupational Safety and Health Administration (OSHA) and the National Fire Protection Association (NFPA) develop standards, such as the National Electric Code (NEC), by which many industries operate. Explosions still occur, but damage can be minimized by using documented protocol for managing them. Companies involved with pharmaceutical development and manufacturing, fuel, chemical manufacturing, food manufacturing, aviation, and other high-risk applications have to abide by these established safety practices.
In each group of flammable-material type (gas/vapor, powder or fiber) the NEC categorizes risky environments based on this basic formula: material’s duration of use + its flammability potential = likelihood for fire. Equipment is designed to
