Choosing and Organizing Cleanroom Furniture to Meet Recommended Gowning Protocol
Manufacturers invest hundreds—even thousands—of dollars per square foot of cleanroom space to meet ISO-proscribed particle counts. Shouldn't the same standards be required of the people who enter and potentially contaminate this ultra-clean environment?
Proper cleanroom garments, including hoods, face masks, booties and gloves, help to contain particles that people emit. Yet improper gowning procedures can negate your investment in cleanliness and threaten yields of sensitive semiconductor devices. Once a garment is contaminated—violated by contact with a dirty surface—it spreads particles everywhere it goes.
You can train personnel on proper garmenting procedures, but how do you guarantee compliance? A violated garment doesn't set off alarms, and few facilities can afford quality control monitors to supervise every person through every washing and dressing stage. Yet if strict controls are not observed, dirty gloves and coveralls almost certainly will come in contact with clean operations.
An Organized Gowning Area that Supports Clean Protocol
The simplest and most economical approach to this dilemma is a correctly designed gowning area, complete with well-designed cleanroom furniture, that keeps personnel on a clean track.
This design starts with a room with pressure greater than that of the outside air, but lower than the cleanroom. This cascading pressure differential reduces the opportunity for contaminants to enter each controlled space. It includes a laminar flow of HEPA-filtered air, typically emitted through ceiling filter/fan units. This continuous wash of clean air immediately removes personnel-emitted contaminants, as well as particles present even in cleanroom-laundered garments (see Figure 1). Failure to maintain the desired particle standard in the change area will lead to contaminating clean garments.
| Activity | No. of particles (>= 0.3µm) |
|---|---|
| Person emits during garmenting process | 3,000,000/min. |
| Cleanest skin (hands) | 10,000,000/ft2 |
| Employee street clothes | 10,000,000 to 30,000,000/ft2 |
| Floor and bench surfaces | > 10,000,000/ft2 |
| Garments supplied by cleanroom laundry | 1,000,000/ ft2 |
| Figure 1: Gowning Area Particle Generators Source: Encyclopedia of Clean Rooms, Bio-Cleanrooms and Aseptic Areas, Dr. Philip Austin, P.E., 2000 | |
The Change Room illustration below depicts an effective floor plan that places the proper gowning stations in appropriate locations. Although many variations on this layout are possible, they should support these key garmenting guidelines:
- Don't Touch! Most cleanliness violations occur when a garment is touched by dirty hands, or when it touches a dirty floor. Hands and gloves should therefore be washed and dried frequently. Select washers and dryers with no-touch IR sensors to minimize the chance of violating gloves. Maintain ultraclean, unviolated surfaces where garments can be donned without contamination. Properly placed gowning benches or use of a special gowning platform helps maintain “not step” areas where a coverall may touch the ground during gowning.
- Dress from the Head Down! That way, released particles won't fall on clean garments or booties.
- Understand Garment Design! In most instances, gloves and booties are worn over coverall sleeves and cuffs, and hoods are tucked inside collars. Make sure that personnel are trained on how to seal these gaps between clothing, and include a mirror to allow final self-inspection. Hang a photo of a correctly garbed person next to the mirror as a "how-to" guide.
- Minimize Motion! As Figure 2 indicates, people wearing cleanroom garments emit thousands of particles per minute even when sitting still! People in street clothes, walking from one change room location to another, release millions of particles. Change room plans should therefore minimize motion, especially during the final stages of dressing, by efficiently organizing the flow of personnel from one station to another.
- Minimize Maintenance! Do you have dedicated, trained personnel to peel off adhesive shoe mats, wipe down gowning benches, keep "clean zones" regularly scrubbed, empty waste receptacles, restock garb dispensers, and perform other upkeep with the absolute regularity that your protocol requires? If not, look for ways to minimize reliance on humans—through automation, careful product positioning, choice of materials, and so forth. Initial cost may be higher, but reduced maintenance and greater compliance provide a rich return on your investment!
- Train and Reinforce! A good change room design supports proper protocol, but it doesn't teach it. Make sure that personnel complete a formal training program, and stress protocol by hanging reminder posters at every gowning station that reinforce proper procedures.
| Personnel Activity | Snap Smock | Standard Coverall | 2-Piece Coverall | Tyvek® Coverall | Membrane Coverall |
|---|---|---|---|---|---|
| No Movement | 100,000 | 10,000 | 4,000 | 1,000 | 10 |
| Light Movement | 500,000 | 50,000 | 20,000 | 5,000 | 50 |
| Heavy Movement | 1,000,000 | 100,000 | 40,000 | 10,000 | 100 |
| Change Position | 2,500,000 | 250,000 | 100,000 | 25,000 | 250 |
| Slow Walk | 5,000,000 | 500,000 | 200,000 | 50,000 | 500 |
| Fast Walk | 10,000,000 | 1,000,000 | 400,000 | 100,000 | 1,000 |
| Figure 2: Austin Contamination Index Particles >= 0.3µm emitted per minute in garment indicated Source: Encyclopedia of Clean Rooms, Bio-Cleanrooms and Aseptic Areas, Dr. Philip Austin, PE, 2000 Note: Light/heavy movement refer to partial body movements (motioning with arm, tapping toes, etc.). Change of position refers to whole body motion (standing up, sitting down, etc.). | |||||
Recommended Gowning Room Design
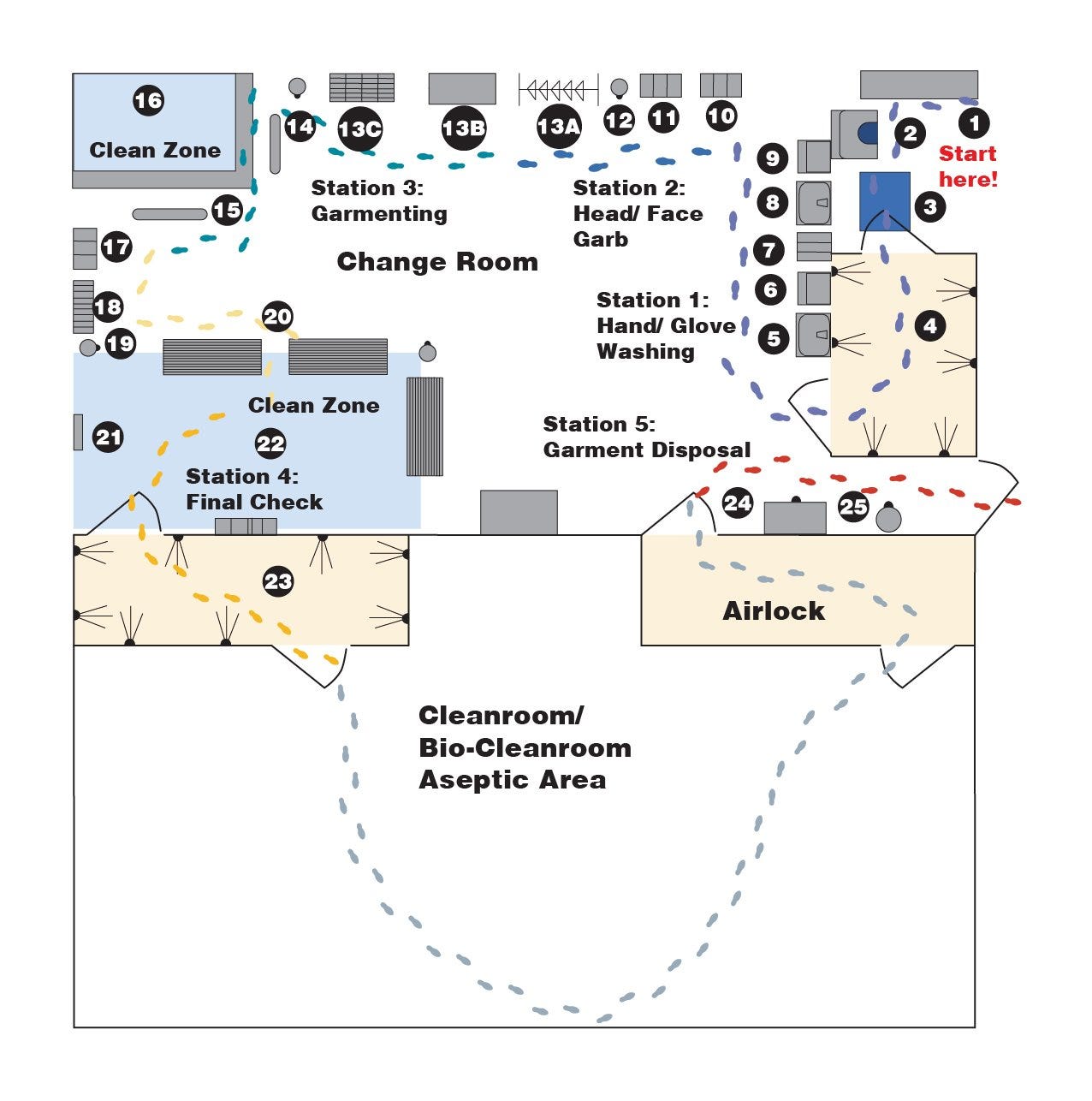
Recommended Cleanroom Furniture
- Secure lockers for clothing and personal property
- Shoe cleaner, with rotating brushes to remove gross contaminants
- Walk-off adhesive Sticky mat
- Air Shower, with HEPA filtered air jets to remove loose contaminants
- Hand washing station, no-touch operation
- Hand dryer (if not incorporated into washer), HEPA/ULPA-filtered air, no touch
- Glove dispenser for glove liner or underglove
- Glove washing/drying station (if gloves not clean)
- Glove washing/drying station (if gloves not clean)
- Headwear dispenser
- Face mask dispenser
- Waste receptacle—should be installed wherever packaging is removed
- Garment Storage Facility—depends on garment type
- Open garment rack for reusable garments
- Enclosed garment cabinet for reusable garments (includes HEPA blower)
- Garment storage shelves for packaged disposable garments, Overstock Cabinet
- Cleanroom table for organization
- Waste receptacle
- Ergonomic lean rails support personnel while donning coveralls
- Gowning platform provides a noncontaminated surface to prevent violation of garment
- Wiper dispenser—used to clean bench prior to putting on booties
- Bootie rack and shoe cover dispenser (may also be incorporated into benches)
- Waste Receptacle
- Gowning benches, used when putting on booties. Booties stay on "clean" side
- Cleanroom mirror for final self-inspection (side-by-side w/poster showing correctly garbed model)
- Safety glass dispenser and overglove dispensers
- Air shower—may be required in cleanroom entrance
- Garment hamper for reusable garments to be laundered
- Waste receptacle for disposable garments

Can't find what you're looking for?Tell us what you need and we'll build it!



















