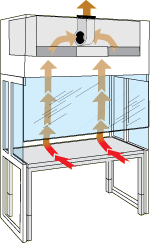
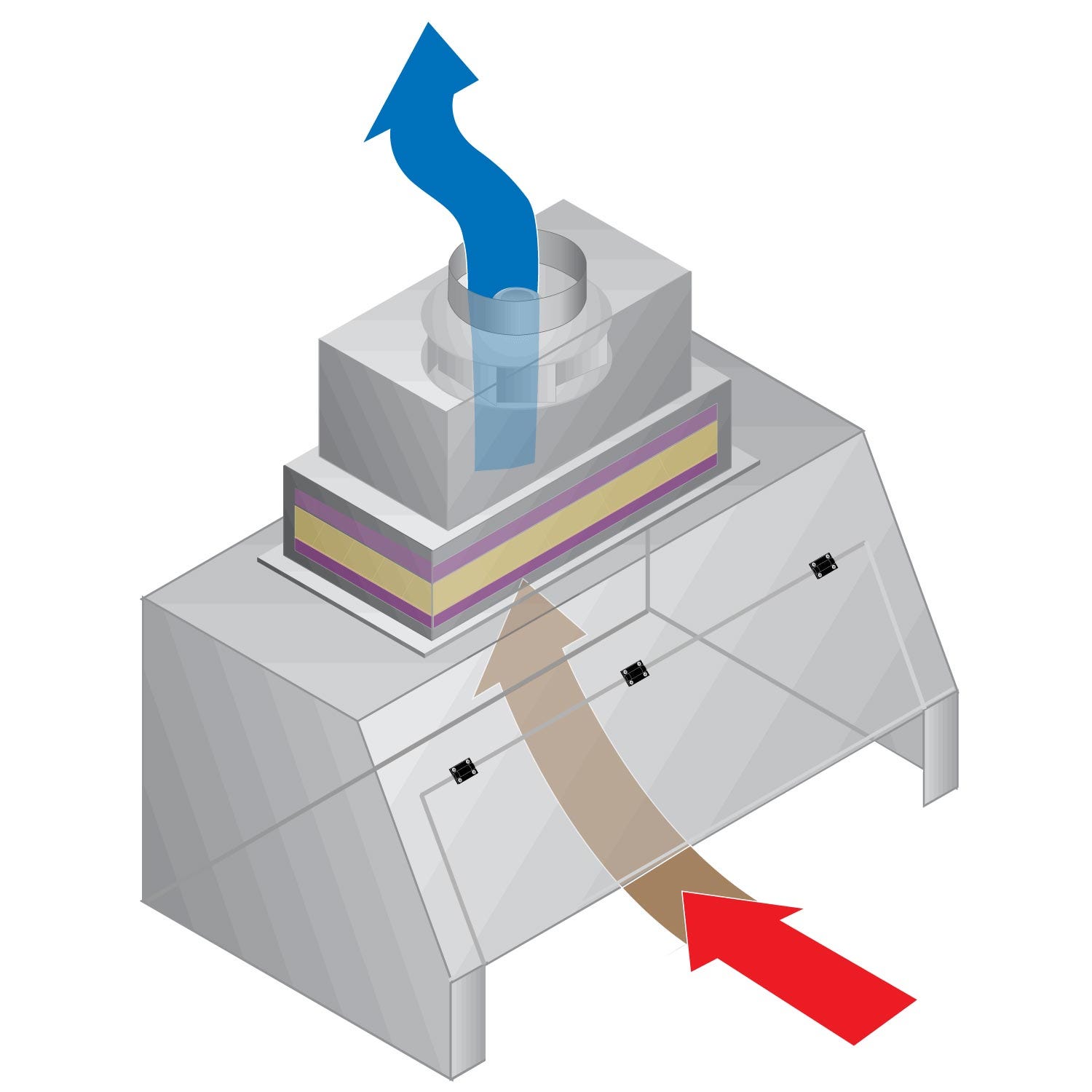
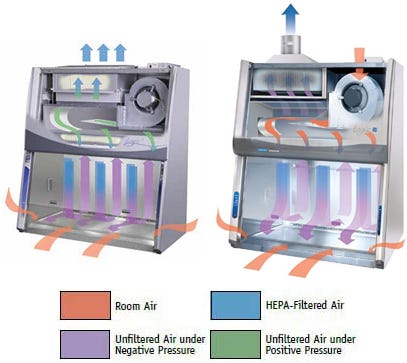
- Available Certified ISO Grade Designs: ISO 5, ISO 6, ISO 7, ISO 8
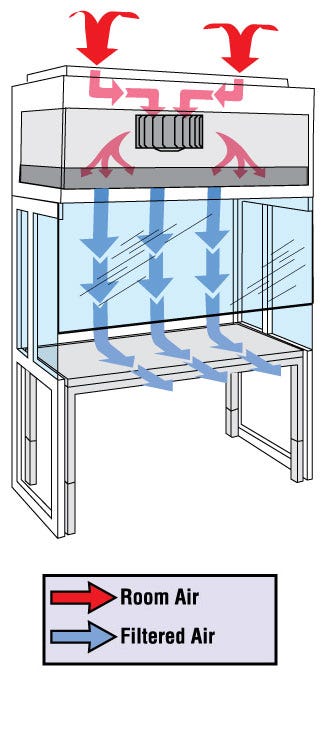
- Bi-directional exhaust system
- Fits above Terra's Wet Processing Station
- Cut outs on the back of the hood for the wet station exhaust flange
- Includes hood, controller, SDPVC side panels, and FFU
- Bench sold separately
• Airflow Design: Vertical
• Depth: 41"
• Hood Design: Floor
• Filter Type: HEPA, RSR
• Frame Material: 304 Stainless Steel
• Height: 90"
• Inside Depth: 32"
• Inside Height: 67"
• Inside Width: 57"
• Light Fixture: Fluorescent
• Manufactured by: Terra Universal
• Max Opening: 32" H
• Model: Wet Processing Station
• Side Panels: Static-Dissipative PVC
• Sash Design: Manual/Sliding
• Sash Height: 35"
• Sash Opening: 16"
• Width: 60"
• Work Area Dimensions: 57" W x 32" D x 67" H
• ISO Rated Air Cleanliness: ISO 5
• Unit of Measure: EA