- +1 (714) 578-6100
Hours Mon - Fri, 07:00 AM - 06:00 PM (Pacific Time)
Biological contaminants are ubiquitous. Microorganisms such as bacteria or fungi roam free in most environments and can cause disease. A major goal of aseptic applications, including surgery, pharmaceutical compounding or medical implant packaging is to prevent contaminants and the potential for disease, so how do we create the ideal setting?
Building a microorganism-free space is exactly what regulatory guidelines aim to achieve. Asepsis is the objective, and certified facilities are bound by regulations that dictate design and protocol for optimizing aseptic processing.
Building the ideal ISO-compliant room consists of a combination of design and materials, which we’ll discuss here, but after the cleanroom is ready for operation, continued success depends on other important issues:
- Establish and execute a robust training program for workers (gowning, work processes, and cleaning techniques, as examples)
- Procured equipment should be properly specified and installed, warrantied and well-maintained
- Cleanroom should consistently pass performance testing of such operational factors as air change rate and particle count
When the cleanroom’s integrity is compromised due to issues like poor control over contaminants or wall seam breaches, time-consuming (i.e., expensive) shutdown, cleaning, re-certification and revalidation are necessary to become operational again. Bottom line: start with a well-designed cleanroom and follow documented procedures to maintain it.
DESIGN
Many regulations list specific requirements for medical, bio or pharmaceutical processes, but also allow some flexibility so that labs can accommodate unique technologies or workflows (reference the USP guidelines for pharmacy compounding, for instance). The regulation will tell you what is necessary; how you choose to enhance the room’s capabilities is between you and your certifier. This section discusses some of the options regarding how the cleanroom will be built and accessorized.
The ability to clean easily, thoroughly and frequently are recurring themes from regulatory agencies. Materials used for rooms and furnishings should resist damage from chemical cleaners (as well as those used in the applications), and be free of obstructions like support braces, seam gaps and protruding hardware. It’s much easier to sanitize a flat, smooth surface than it is one with angles and corners. Particles are small; they’ll take any opportunity to find a hiding place.
One design strategy is coved corners along floors and walls that dispense with hard-to-clean right angles. Another would be meticulous seam sealing of wall and ceiling panels, using a cleanroom-compatible sealant.

Image 1: Stainless steel corner coving with FRP wall panels.
Paneling materials such as fiberglass-reinforced plastic (FRP) and chlorinated polyvinyl chloride (C-PVC) can be adhered to gypsum wallboard or concrete walls, giving you a choice between creating a modular room or converting existing office space into a cleanroom. Ceiling grids that house FFUs, lights and other modules can be suspended or supported above these walls, completing the enclosed interior. Both design options are less expensive than constructing a new cleanroom. Modular rooms give you the flexibility to expand, scale-down or relocate, so may be preferred over existing construction.

Image 2: FRP installation in progress.

Image 3: FRP installation complete; room in ”as built” condition.
These before/after photos of wall ducts (Image 4) along the floor show the process of sealing the cracks and gaps when using FRP wall panels.

Steel ceiling grids for BioSafe® rooms are easy to wipe down for cleaning, and support all necessary modules, including HEPA or ULPA fan/filter units, fluorescent lighting, ceiling panels and options such as UV-C sterilizers and ionizing bars for neutralizing electro-static discharges (ESD). These ceiling grids top off free-standing modular rooms, or can be attached to existing facility walls when a room conversion is preferred.
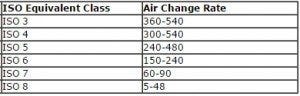 Room-side replaceable fan/filter units are ideal for aseptic rooms. Filters can be replaced from inside the cleanroom, without breaching the enclosure. Because they eliminates the need for roof access, this reduces maintenance time and cost. Don’t forget to calculate your FFU ceiling coverage based on room size and ISO level, then test your air change rates to be in compliance.
Room-side replaceable fan/filter units are ideal for aseptic rooms. Filters can be replaced from inside the cleanroom, without breaching the enclosure. Because they eliminates the need for roof access, this reduces maintenance time and cost. Don’t forget to calculate your FFU ceiling coverage based on room size and ISO level, then test your air change rates to be in compliance.
MATERIALS
As with so many other projects, the devil’s in the details. For example, you might consider installing an ultraviolet-C module in the ducting system: expose the airstream to radiation, killing airborne bacteria and viruses before they reach the FFUs. Other considerations for efficient cleanroom design? Try flush-mounted pass-throughs and windows that reduce the surface area from ledges, making cleaning easy and fast.
Now that you have some ideas about the design of the ISO 5 – 8 aseptic cleanroom, you should consider your building materials options. The first is steel. Terra’s steel BioSafe rooms come in powder-coated steel, or 304 or 316 stainless steel. The highest grade is 316; its cost is greater, but so is its resistance to corrosion. Stainless steel is made even cleaner by electro-polishing, a reverse plating process that removes metal impurities using electricity and chemicals. An ultra-smooth surface is the result: fewer peaks and valleys where particulates can hide.

Image 5: Stainless steel aseptic cleanroom.
Plastic wall panels made of FRP or C-PVC is another choice. Seams in-between FRP panels are closed off with sealants approved for use in cleanrooms, and have the added benefit of expanding or contracting to accommodate temperature-induced changes without cracking. FRP panels are available as Class A or Class C; the material is similar, but the Class A is Factory Mutual (FM) approved for facilities that need materials with low flame spread due to work with flammable chemicals or supplies. These FRP panels have a patented surfacing technology that ensures a uniform seal to enhance durability, prevent generation of particles and suppress microbial growth.

Image 6: Cleanroom wall and ceiling space covered with C-PVC paneling.
C-PVC panel seams are welded to create uniform surfaces where germs and contaminants won’t gather, and their unique solvent-fee silicone adhesive allows installation on most wall surfaces. C-PVC panels are also FM rated and certified to achieve ISO 5 cleanliness. These compressed panels offer excellent chemical resistance and prevent particle emission and microbial growth.
All of these all panel options share two important characteristics:
- They do not contribute to the population of contaminants in the room. They are non-shedding, non-out-gassing and scratch-resistant
- Most common cleaners can be used to sterilize surfaces; they are resistant to damage from chemicals such as isopropyl alcohol (IPA), and stand up to the rigors of hydrogen peroxide vapor and UV sterilization
CONCLUSION
Aseptic materials and room design options help companies prepare to perform important medical or bio/pharmaceutical operations, as well as others requiring an easy-to-sterilize environment. You can get such a room in a few days and be ready for operation quickly, while meeting regulatory guidelines. Choices presented here should be weighed against budget, applications, facility limitations and preference. Learn more about Terra’s cleanroom designs and materials here.
Terra Universal is the leading expert in the design and fabrication of ISO rated cleanrooms, furnishing and supplies.
Get a free consultation from one of our cleanroom specialists:
Call (714) 459-0731



